
News Archive
You will find the latest information about our company and industries here. Please feel free to contact us with any questions and suggestions relating to our topics or content!

Wasserung Rotomarin
Today our dynamic test rig "RotoMarin®" was launched. It was developed by the Institute for Antifouling and Biocorrosion to investigate the growth of fouling on surfaces under the most realistic conditions possible. For this purpose, the surfaces to be examined are screwed as circular segment-shaped test plates onto plates moved by a rotating axis and moved at speeds of up to 19 knots.

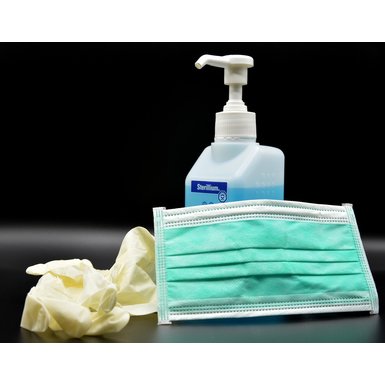
Online Update Hygiene and infection prevention: COVID-19
The Corona Pandemic poses major challenges for all of us. The HIHeal network (https://hiheal.de/home.html) is launching a free series of webinars to provide information on important issues related to the topic.
The first webinar will take place on 26.05.2020 from 10:00 - 12:00.
Further information: https://hiheal.de/news/online-update-hygiene-und-infektionspraevention-c...

Support for local production of disinfectants and face masks in Africa
In order to stem the spread of the corona virus in Malawi, Dr. Brill + Partner GmbH together with "tat für tat malawi e.V." supports the local production of disinfectants and face masks in Malawi. Through an appeal for donations, more than 4,000 € have been collected. The production in Malawi is in the process of preparation.
EU Commission: Introduction of medical devices regulation postponed
At the end of May, according to the new EU Medical Devices Regulation, only specially certified medical devices should actually be placed on the market in the European Union. The regulation should now enter into force one year later in order to avoid creating additional bureaucratic hurdles in the corona crisis. Further details: https://ec.europa.eu/germany/news/20200325-corona-medizinprodukte_de

Successful candidacy: Dr. Florian Brill elected to the Plenum of the Hamburg Chamber of Commerce
In the 2020 plenary election of the Hamburg Chamber of Commerce, the electoral alliance Starke Wirtschaft Hamburg, of which Dr. Florian Brill was also a member, was able to win 71 % of all votes. Dr. Florian Brill successfully ran in the electoral group "Services" for medium-sized companies and will now represent his goals in the plenary session of the Chamber of Commerce. Dr. Brill and the entire electoral alliance "Starke Wirtschaft Hamburg" would like to thank you for the trust you have placed in us and for your great support over the past few weeks. Dr. Brill said about his motivation before the election: "My goals are to reunite the chamber in order to achieve that honorary and full-time office work together again in an optimal way. I would like the Chamber of Commerce to be perceived again as the voice of Hamburg's economy, loud and clear. This is important in order to tackle the challenges facing the City of Hamburg on the issues of digital transformation, the shortage of skilled workers and the dilemma of "transport space" vs. "commercial space" vs. "living space" for a golden future for the city. SMEs in particular need a chamber of commerce with a 360° view, so that it can be at your side in all phases, from start-up, through transformation, innovation and the development of new business fields, internationalisation, and the training and further education of skilled workers. This is what I will fight for, always taking the values of a family entrepreneur and VEEK as my foundation. So that Hamburg becomes even more successful." https://www.facebook.com/StarkeWirtschaftHamburg/posts/187508182650236

New research project on antifouling
New research project for the certification of biocide-free antifouling systems with the Blue Angel
On behalf of the Federal Environment Agency, Hydrotox and the Institute for Antifouling and Biocorrosion at Dr. Brill + Partner GmbH will be working on a further research project from September 2019 to August 2021, in which award criteria for 1 to 2 environmental labels (Blue Angel) for biocide-free antifouling products will be developed.

Website-Relaunch
Wir haben unsere Website überarbeitet und verschiedene neue Bereiche kreiert, die die Informationen noch besser und einfacher transportieren werden als. Natürlich hat auch unser neues Unternehmen Dr. Brill + OBJEKTuS GmbH & Co. KG Institut für Hygiene und Mikrobiologie eine neue Heimat bekommen, die wir ständig weiterentwickeln. Viel Spaß auf unserer Seite.

Fusion! Dr. Brill + Objektus GmbH & Co. KG Institut für Hygiene und Mikrobiologie

CAHMV 2018 - Register now!

MEDICA 2018 and Arab Health 2019

Extension of the scope of accreditation in the virucidal field
